Lab Description
The Peri-Operative Neurocognitive Research Team (PORT) Laboratory studies neurocognitive function in older adults, particularly in the setting of major surgery. We are especially interested in the interaction between patient factors (including neurodegenerative pathology), perioperative stressors, postoperative delirium, longer term cognitive and physical function, and the risk of developing Alzheimer’s disease and related dementias. PORT lab members and collaborators have training in a variety of fields ranging from clinical anesthesiology, neuropsychology and neuroimaging, cognitive neuroscience, sleep medicine, biostatistics, clinical research, flow cytometry, proteomics, to mouse genetics.
Values: Discovery, Collaboration, Honesty, Integrity, Humility, Transdisciplinary Approaches, Innovation, Mentorship, Collegiality, and Fun.
Major Research Interests
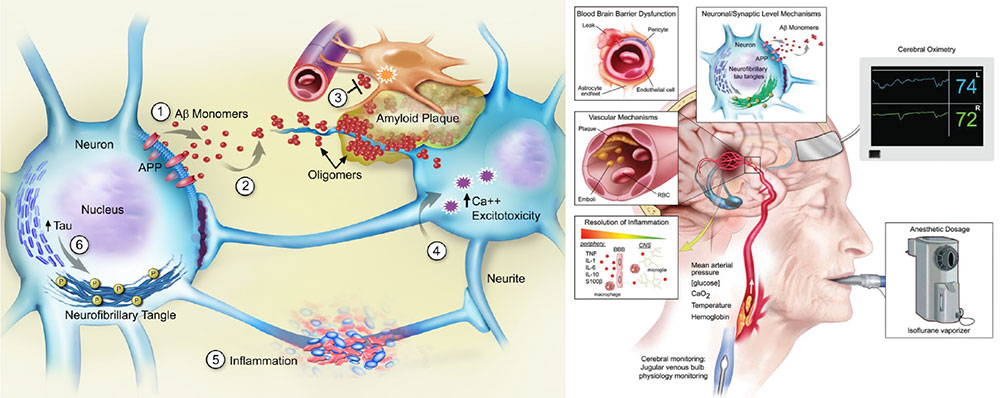
The Systems Neuroscience of Perioperative Neurocognitive Disorders: In work led by Dr. Joseph P. Mathew and Dr. Jeffrey Browndyke, our group has performed several of the first functional neuroimaging studies of postoperative cognitive dysfunction, utilizing resting- and task-based functional magnetic resonance imaging (fMRI) in older (age >60) adults. Our work, now confirmed by independent research labs in the field, suggests possible dysregulation of the brain’s default mode network (DMN) is associated with the severity of cognitive dysfunction 6 weeks after cardiac surgery in older adults. We have found reduced degree centrality of the posterior cingulate cortex, a major DMN “hub,” suggesting reduced intranetwork coherence in patients with postoperative cognitive dysfunction. This DMN “hub” paradoxically demonstrates reduced anticorrelation during cognitive task performance in patients with postoperative cognitive dysfunction, suggesting that poor task performance in these patients may be due, in part, to dynamic brain inter-network dysregulation. For more information on this work, please see https://pubmed.ncbi.nlm.nih.gov/27858963/ and https://pubmed.ncbi.nlm.nih.gov/29164774/. Additional and ongoing laboratory studies in these cardiac and non-cardiac surgical patients are examining the structural and functional neuroimaging variable associations with perioperative biomarker changes and more granular changes in cognition and mood after surgery.
A large body of work in recent years has described specific electroencephalography (EEG) patterns detectable on the scalp surface in response to distinct anesthetic drugs (such as propofol and the volatile anesthetics, among others). Key work by Patrick Purdon and colleagues demonstrated that EEG alpha (i.e. 8-12 Hz) signal amplitude (or power) declines with age in older adults. Our group was then the first to show that older adults have significant variability in alpha oscillation power in response to propofol and the inhaled volatile anesthetics, and that the magnitude of frontal EEG alpha oscillation power correlates with baseline/preoperative cognitive function in older adults. We found this result in two different cohorts using both the raw EEG signal from a proprietary BIS monitor, and the EEG data from the frontal electrodes of a 32 channel EEG electrode array. For more on this work, please see https://pubmed.ncbi.nlm.nih.gov/28533746/.
In parallel with this line of work, we have also examined anesthetic dosage and brain monitoring practices as a function of increasing. It has long been none that the minimum alveolar concentration (MAC) of the volatile inhaled anesthetics declines by age; our recent systematic literature search and meta-regression analysis demonstrated that MAC declines by 6.47% per decade after 1 year of age, with an R2 value of 0.98 (https://www.sciencedirect.com/science/article/pii/S000709121930755X?via%3Dihub). We hypothesized that clinicians at a major academic medical center would decrease the amount of volatile inhaled anesthetics delivered to older adults by less than this 6.47% decrease per decade, which would result in a relative increase in volatile inhaled anesthetic dosage in older adults. In a study of over 17,000 patients, we found that this hypothesis was correct. Yet, we found that a processed EEG measure of so-called “anesthetic depth” (the BIS index) actually increased in older adults, despite the fact that higher anesthetic dosage in older adults would be predicted to result in lower BIS numbers. For more information on this “paradox of age”, please see https://pubmed.ncbi.nlm.nih.gov/31279479/. This work suggests the need for better EEG monitors for use in clinical practice on older adults undergoing general anesthesia, and that more careful anesthetic titration in older adults could reduce dose-dependent side effects of the inhaled volatile anesthetics and propofol.
The Relationship between Alzheimer’s Disease, Anesthesia and Surgery, and Human Perioperative Neurocognitive Disorders: Numerous investigators have theorized that there may be a link between human perioperative neurocognitive disorders and Alzheimer’s Disease and Related Dementias (ADRD). We know that patients with preclinical ADRD are at increased risk for developing postoperative delirium and cognitive dysfunction, and patients who develop these perioperative neurocognitive disorders develop ADRD at increased rates. We have found that there are significant increases in CSF levels of the microtubule associated protein tau (a neural injury biomarker) at 10 and 24 hours following surgery in old adults, and these was no difference in these CSF tau increases among patients whom we randomized to propofol versus isoflurane for anesthesia maintenance. For more on this work, please see https://pubmed.ncbi.nlm.nih.gov/27079717/.
To determine whether the magnitude of these postoperative CSF tau increases are associated with the severity of postoperative cognitive dysfunction, we then initiated the Markers of Alzheimer’s Disease and neuroCognitive Outcomes following Perioperative Care (MADCO-PC) cohort study in older adults (manuscripts under preparation). The MADCO-PC study also examines whether postoperative CSF tau increases are associated with postoperative changes in functional brain connectivity (as measured by fMRI).
The Role of Neuroinflammation in Human Perioperative Neurocognitive Disorders: Several groups (including our own) have found that anesthesia and surgery are associated with an increase in inflammation within the central nervous system. We were the first to demonstrate a possible influx of peripheral monocytes into the cerebrospinal fluid following major surgery in older adults (https://pubmed.ncbi.nlm.nih.gov/31085945/). We have also found significant increases in pro-inflammatory cytokines within human CSF following surgery (https://pubmed.ncbi.nlm.nih.gov/29181002/). We are currently examining the role of both cellular and molecular markers of inflammation in the pathogenesis of human perioperative neurocognitive disorders and associated brain functional connectivity changes in both the INTUIT and MARBLEstudies. INTUIT stands for Investigating NeuroinflammaTion Underlying postoperative neurocognITive and MARBLE stands for Modulating ApoE to Reduce Brain inflammation, deLirium and cognitivE dysfunction after surgery in older adults. For more on the INTUIT study please see our study methods paper at https://pubmed.ncbi.nlm.nih.gov/30674067/ and our study registration page at https://www.clinicaltrials.gov/ct2/show/NCT03273335?term=INTUIT&draw=2&rank=2. For more information about the MARBLE study, please see our study methods paper at https://pubmed.ncbi.nlm.nih.gov/32417770/ and our study registration page at https://www.clinicaltrials.gov/ct2/show/NCT03802396?term=MARBLE+Berger&draw=2&rank=1.
Methods/Techniques
- Functional, Structural MRI
- Electroencephalography (EEG)
- Cognitive Testing
- Delirium Screening
- Physical Function Assessments
- Quality of Life, Mental Health Assessments
- Polysomnography (Sleep studies)
- CSF Studies
- Genetics
- Proteomics
Lab Members
Postdoctoral Fellows
Tyler Reekes, PhD
Postdoctoral Fellow
Melody Reese, PhD
Postdoctoral Fellow
Clinical Research Staff
Melissa Adams, BS
Clinical Research Specialist, Sr.
Micheal Bennett, BS
Clinical Research Specialist, Sr.
Piper Boykin, BS
Clinical Research Coordinator
Rachele Brassard, BS
Clinical Research Specialist
Morgan DePrizio, BS
Clinical Research Specialist, Sr.
Lori Cole-Freiburger
Clinical Research Specialist
Refaat Hassan, MB BChir, MA
Research Scholar
Reid Funderburk, BS
Clinical Research Specialist, Sr.
Courtney Holland, BS
Clinical Research Coordinator
Bethany Hsia, PhD
Research Program Leader
Macy Le, MSc
Clinical Research Specialist, Sr.
Christy Peterson, BS
Clinical Research Coordinator
Jamarc Simon, BA
Clinical Research Coordinator
Noah Timko, MPH
Clinical Research Coordinator
Yanne Toulgoat-Dubois, BS
Clinical Research Coordinator
Undergraduate Students
Jack Fallon
Duke Class of 2024
Brendan Kelleher
Duke Class of 2024
Sydney Maynor
Duke Class of 2025
Vinith Upadhya
Duke Class of 2026
Jonathan Valdes
Duke Class of 2025
Sophie Wu
Duke Class of 2024
Local Collaborators
Mary Cooter, MS
Biostatistician
Duke, Department of Anesthesiology
Duke Immune Profiling Core
Drs. Kent Weinhold, Janet Staats, Jennifer Enzor, Twan Weaver
Duke Molecular Physiology Institute
Janet Huebner, MS and Mike Muehlbauer, PhD
Duke Proteomics Core Facility
Arthur Moseley, J. Will Thompson, Matthew Foster
Dr. Joseph P. Mathew
Jerry Reves Professor & Chair
Duke, Department of Anesthesiology
Dr. Andrew Spector
Sleep Medicine Specialist
Duke, Department of Neurology
Dr. Niccolo Terrando
Associate Professor
Duke, Department of Anesthesiology
Dr. Heather Whitson
Duke Center for the Study of Aging & Human Development
Duke, Department of Medicine, Geriatrics Division
Dr. Marty Woldorff
Duke Center for Cognitive Neuroscience & Department of Psychology & Neuroscience
Current External Collaborators
Courtney Balentine, MD, MPH
Associate Professor
Endocrine Surgery Division
University of Wisconsin
Joseph Lucas, PhD
Statistician
Vital Statistics, LLC
Chapel Hill, NC
Dr. John Newman
UCSF/Buck Institute
Bay Area, CA
Dr. Beth Stevens
Harvard/MIT Broad Institute
Harvard Medical School
Boston, MA
Dr. Brandon Westover
Massachusetts General/Brigham Hospital
Harvard Medical School
Boston, MA
Dr. Henrik Zetterberg
University of Gothenberg
Sweden
In the News
2022
Congratulations to medical student, Daniel Villalobos, on winning the Clinical Research Award for his project, “Perioperative Neurocognitive and CSF Alzheimer's Biomarker Trajectories in Older Patients Randomized to Isoflurane vs Propofol for Anesthetic Maintenance,” at Duke’s 5th Annual Office of Physician Scientist Development Symposium. Villalobos is a part of Dr. Miles Berger’s Peri-Operative Neurocognitive Research Team Laboratory. (March 24, 2022)
Congratulations to medical student, Megan Wong, on receiving the Kosaka Best Abstract Award in the clinical research category at the 2022 International Anesthesia Research Society Annual Meeting for her project, titled “Cognitive and cerebrospinal fluid Alzheimer’s Disease biomarker changes over time in older surgical patients and matched nonsurgical controls.” (March 18-20, 2022)
2021
A new study, led by Duke Anesthesiology’s Dr. Miles Berger, finds that people with an Alzheimer’s risk gene have altered inflammatory protein levels in their nervous system that are detectable years before symptoms arise. READ MORE
2020
Congratulations to Dr. Michael Devinney from the PORT lab for winning a Dream Innovation Grant (DIG) from the Duke Anesthesiology Department for his project “Proteomic Determination of Neuroinflammation in Postoperative Delirium.
Dr. Devinney was also recently received a 2020 George Maddox Award from the Duke Center for the Study of Aging & Human Development.
Dr. Devinney also received the 2020 William H. Young Neuroscience Research Award from the Society for Neuroscience in Anesthesiology & Critical Care.
Dr. Miles Berger of the PORT lab recently gave a lecture for the American Society of Anesthesiology Perioperative Brain Health Initiative Virtual Town Hall on “Intraoperative EEG to Identify Preoperative Neurocognitive Vulnerability”
Dr. Browndyke of the PORT Lab recently was interviewed by Anesthesiology for their podcast series regarding breaking studies suggesting that postoperative delirium and postoperative cognitive decline may be distinct pathological conditions.
Selected Publications
- VanDusen KW, Li YJ, Cai V, Hall A, Hiles S, Will Thompson J, Arthur Moseley M, Cooter M, Acker L, Levy JH, Ghadimi K, Quiñones QJ, Devinney MJ, Chung S, Terrando N, Moretti EW, Browndyke JN, Mathew JP, Berger M; MADCO-PC Investigators. Cerebrospinal Fluid Proteome Changes in Older Non-Cardiac Surgical Patients with Postoperative Cognitive Dysfunction. J Alzheimers Dis. 2021 Feb 26.
- Nobuhara CK, Berger M. Risk of Spinal Hematoma After Lumbar Puncture. JAMA. 2021 Feb 23;325(8):787-788.
- Terrando N, Park JJ, Devinney M, Chan C, Cooter M, Avasarala P, Mathew JP, Quinones QJ, Maddipati KR, Berger M. Immunomodulatory lipid mediator profiling of cerebrospinal fluid following surgery in older adults. Sci Rep. 2021 Feb 4;11(1):3047.
- Balentine CJ, Meier J, Berger M, Reisch J, Cullum M, Lee SC, Skinner CS, Brown CJ. Using Local Anesthesia for Inguinal Hernia Repair Reduces Complications in Older Patients. J Surg Res. 2021 Feb;258:64-72.
- Meier J, Berger M, Hogan T, Reisch J, Zeh H, Cullum CM, Lee SC, Skinner CS, Brown CJ, Balentine CJ. Using local rather than general anesthesia for inguinal hernia repair may significantly reduce complications for frail Veterans. Am J Surg. 2021 Jan 22.
- Velkers C, Berger M, Gill SS, Eckenhoff R, Stuart H, Whitehead M, Austin PC, Rochon PA, Seitz D. Association Between Exposure to General Versus Regional Anesthesia and Risk of Dementia in Older Adults. J Am Geriatr Soc. 2021 Jan;69(1):58-67.
- Berger M, Cooter M, Roesler AS, Chung S, Park J, Modliszewski JL, VanDusen KW, Thompson JW, Moseley A, Devinney MJ, Smani S, Hall A, Cai V, Browndyke JN, Lutz MW, Corcoran DL. APOE4 Copy Number-Dependent Proteomic Changes in the Cerebrospinal Fluid. J Alzheimers Dis. 2021;79(2):511-530.
- Oyeyemi DM, Cooter M, Chung S, Whitson HE, Browndyke JN, Devinney MJ, Smith PJ, Garrigues GE, Moretti E, Moul JW, Cohen HJ, Mathew JP, Berger M. Relationship Between Depression/Anxiety and Cognitive Function Before and 6 Weeks After Major Non-Cardiac Surgery in Older Adults. J Geriatr Psychiatry Neurol. 2020 Dec 30;:891988720978791.
- Berger M, Ghadimi K. Necessary Heroes and Ethos, from Fighting Nazis to COVID-19. Anesthesiology. 2020 Dec 1;133(6):1307-1310.
- Balentine CJ, Meier J, Berger M, Hogan TP, Reisch J, Cullum M, Zeh H, Lee SC, Skinner CS, Brown CJ. Using local rather than general anesthesia for inguinal hernia repair is associated with shorter operative time and enhanced postoperative recovery. Am J Surg. 2020 Aug 25.
- Vasunilashorn SM, Devinney MJ, Acker L, Jung Y, Ngo L, Cooter M, Huang R, Marcantonio ER, Berger M. A New Severity Scoring Scale for the 3-Minute Confusion Assessment Method (3D-CAM). J Am Geriatr Soc. 2020 Aug;68(8):1874-1876.
- Nobuhara CK, Bullock WM, Bunning T, Colin B, Cooter M, Devinney MJ, Ferrandino MN, Gadsden J, Garrigues G, Habib AS, Moretti E, Moul J, Ohlendorf B, Sandler A, Scheri R, Sharma B, Thomas JP, Young C, Mathew JP, Berger M. A protocol to reduce self-reported pain scores and adverse events following lumbar punctures in older adults. J Neurol. 2020 Jul;267(7):2002-2006.
- Klinger RY, Bottiger B, Berger M. A Plague on Both Your Monitors! Are Bispectral Index and Cerebral Oximetry Useful for Reducing Postoperative Cognitive Impairment?. J Cardiothorac Vasc Anesth. 2020 May;34(5):1182-1183.
- Wang P, Velagapudi R, Kong C, Rodriguiz RM, Wetsel WC, Yang T, Berger M, Gelbard HA, Colton CA, Terrando N. Neurovascular and immune mechanisms that regulate postoperative delirium superimposed on dementia. Alzheimers Dement. 2020 May;16(5):734-749.
- Berger M, Mark JB, Kreuzer M. Of Parachutes, Speedometers, and EEG: What Evidence Do We Need to Use Devices and Monitors?. Anesth Analg. 2020 May;130(5):1274-1277.
- Berger M, Cooter M, Ni K, Thomas J, Gupta DK, Kertai MD. Response to ‘Correlation between bispectral index and age-adjusted minimal alveolar concentration’ (Br J Anaesth 2020; 124:e8). Br J Anaesth. 2020 Mar;124(3):e38-e39.
- Cooter M, Ni K, Thomas J, Gupta DK, Hopkins TJ, Miller TE, James ML, Kertai MD, Berger M. Age-dependent decrease in minimum alveolar concentration of inhaled anaesthetics: a systematic search of published studies and meta-regression analysis. Br J Anaesth. 2020 Jan;124(1):e4-e7.
- VanDusen KW, Eleswarpu S, Moretti EW, Devinney MJ, Crabtree DM, Laskowitz DT, Woldorff MG, Roberts KC, Whittle J, Browndyke JN, Cooter M, Rockhold FW, Anakwenze O, Bolognesi MP, Easley ME, Ferrandino MN, Jiranek WA, Berger M. The MARBLE Study Protocol: Modulating ApoE Signaling to Reduce Brain Inflammation, DeLirium, and PostopErative Cognitive Dysfunction. J Alzheimers Dis. 2020;75(4):1319-1328.
Photos





Resources
- Duke Office of Clinical Research
- Duke Office of Regulatory Affairs & Quality
- Duke IRB
- Brain Imaging and Analysis Center (BIAC)
- Society for Neuroscience in Anesthesiology and Critical Care (SNACC)
- International Anesthesia Research Society (IARS)
- Foundation for Anesthesia Education and Research (FAER)
- Alzheimer’s Association International Conference (AAIC)
- Early-Stage Anesthesiology Scholars (eSAS)




